Symposium Proceedings Published



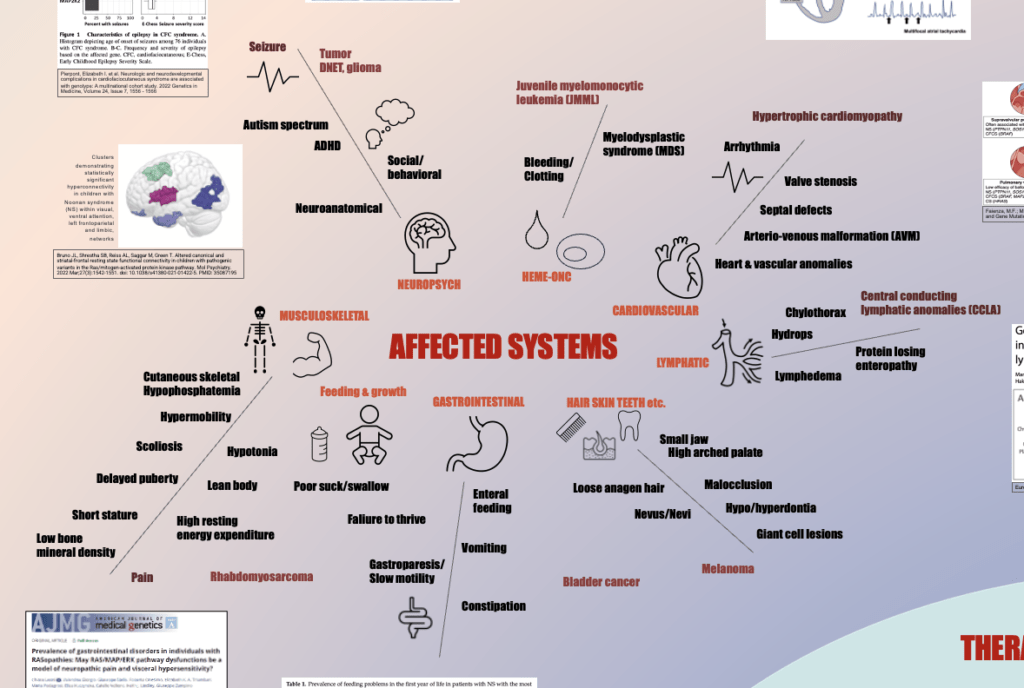
ABSTRACT The RASopathies are a group of congenital disorders with overlapping clinical manifestations that are…
Beth StronachMarch 17, 2026